- Guidance
ISO 10993-6
- Animals
Rat, Rabbit
ISO 10993 Biocompatibility Tests
Biocompatibility testing is a key step in medical device development, ensuring that products are safe under their intended clinical use. Through rigorous biological risk identification, these evaluations assess how a device interacts with the human body in advance, enabling effective control of potential biological hazards prior to market release.
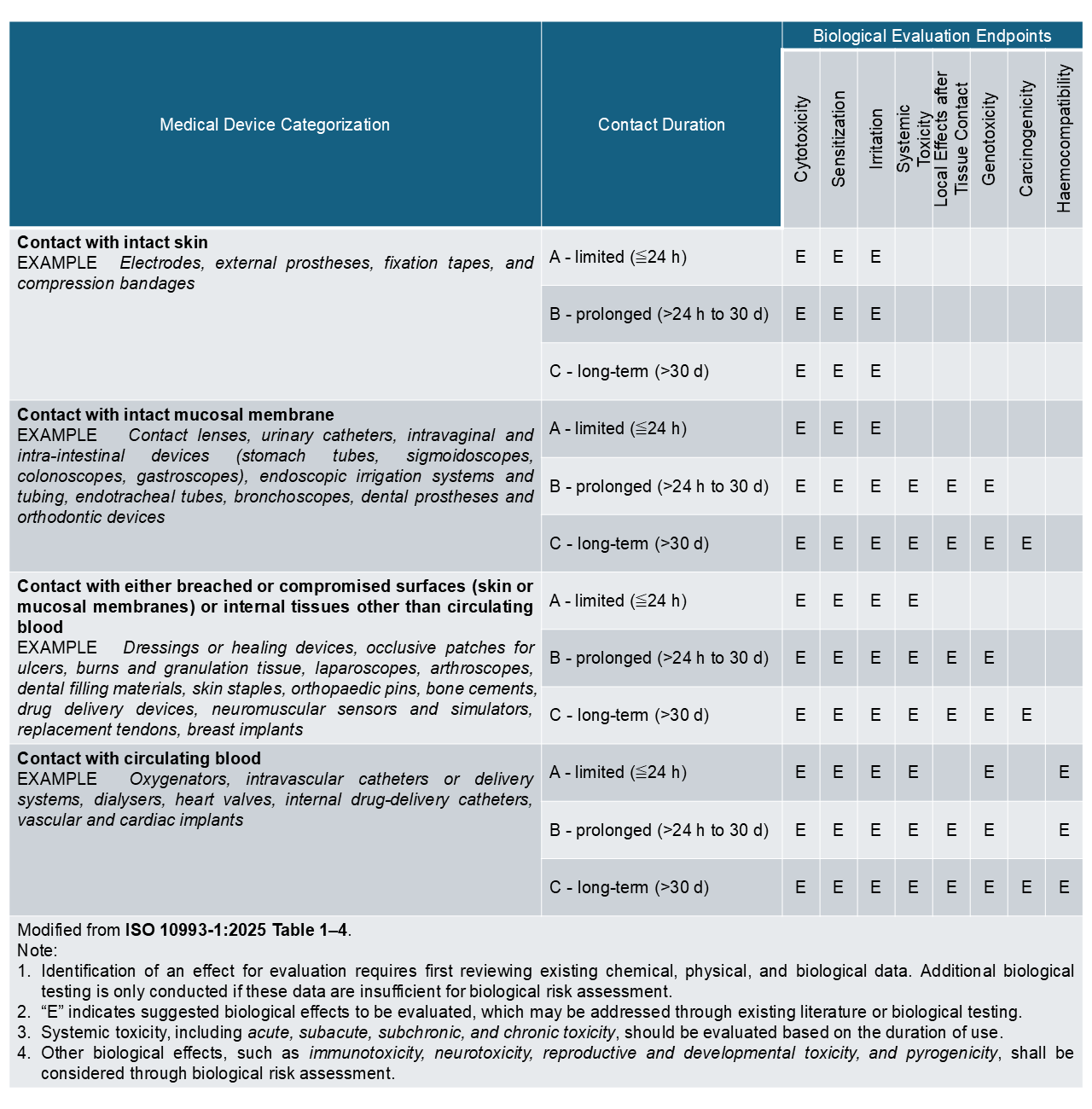
The “Big Three” tests, cytotoxicity, irritation, and sensitization, represent the minimum standard for biological safety evaluation for most medical devices. Depending on the device’s materials, intended use, and the type and duration of body contact, additional endpoints may be required to evaluate biological effects. These may include systemic toxicity, local effects after tissue contact, genotoxicity, carcinogenicity, and haemocompatibility assessments. Biological evaluation strategies are guided by ISO 10993-1, which provides a risk-based framework based on the nature and duration of body contact (see evaluation matrix below).
At Medgaea, we offer scientifically rigorous biocompatibility strategies, delivered by experienced professionals in globally accredited laboratory facilities, that meet the requirements of regulatory authorities worldwide.